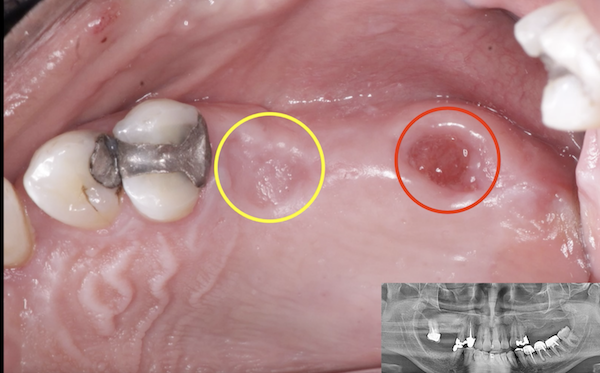
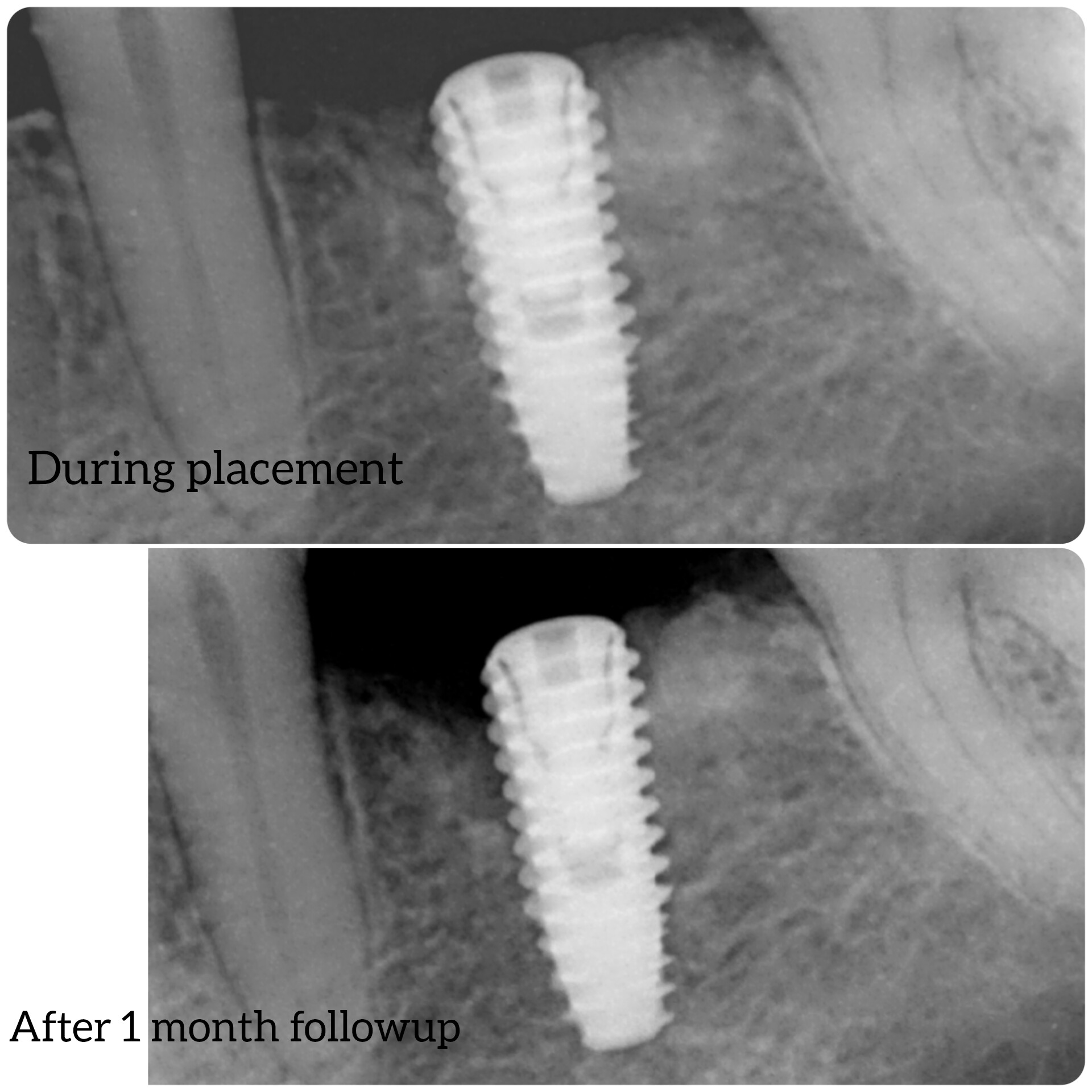
NuOss, is deproteinized, bovine origin, cancellous bone ( (Xenograft). And just like allografts ( from Human) they have the disadvantage that their preparation during freeze drying and irradiating lowers the materials Osteogenic powers,And the body’s response to it ( Immunological) may lesson in terms of incorporating it to bone.
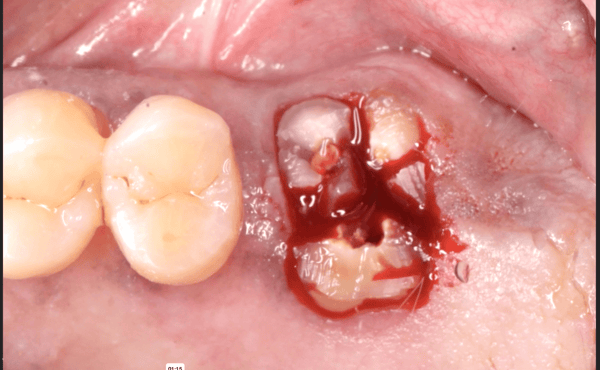
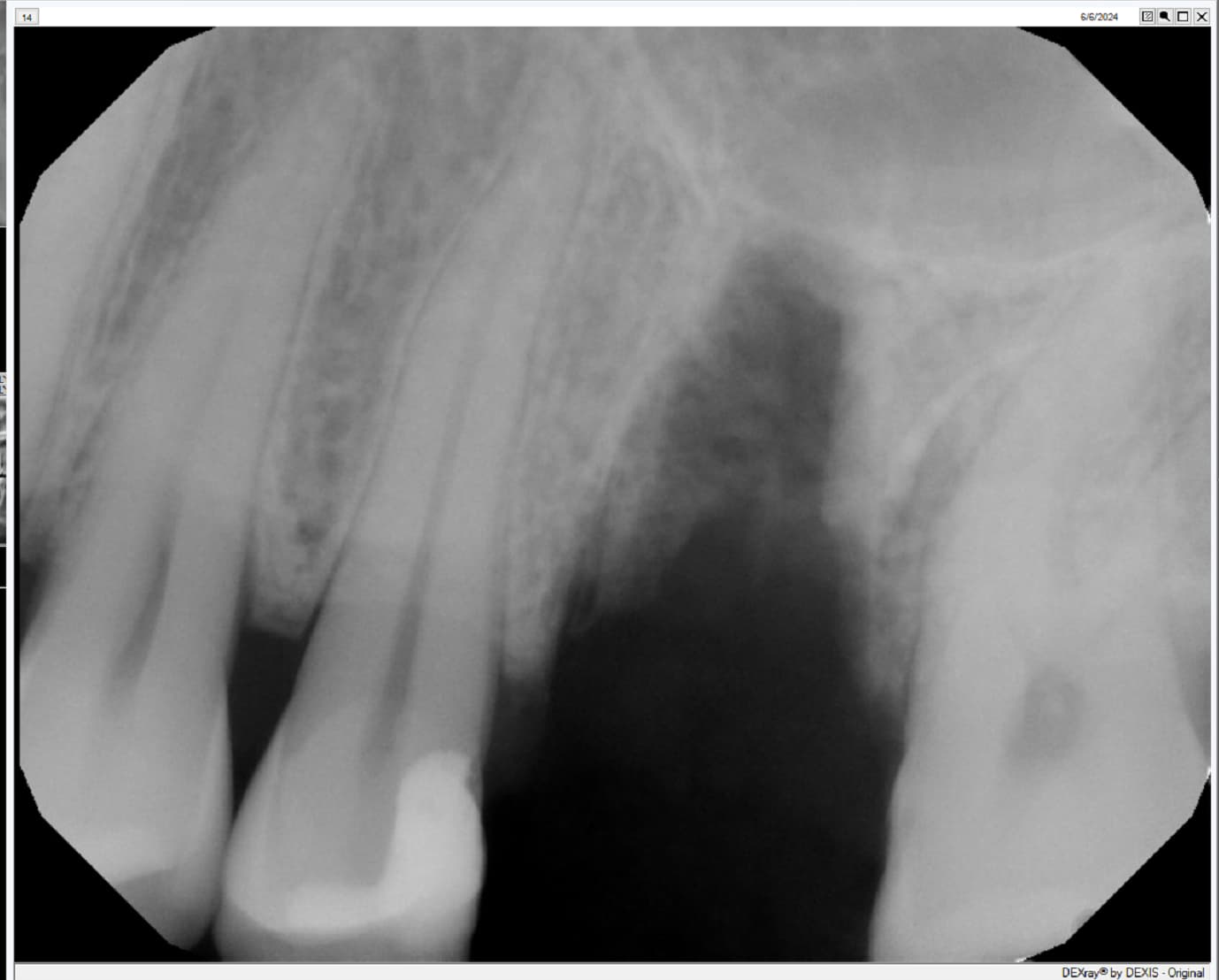
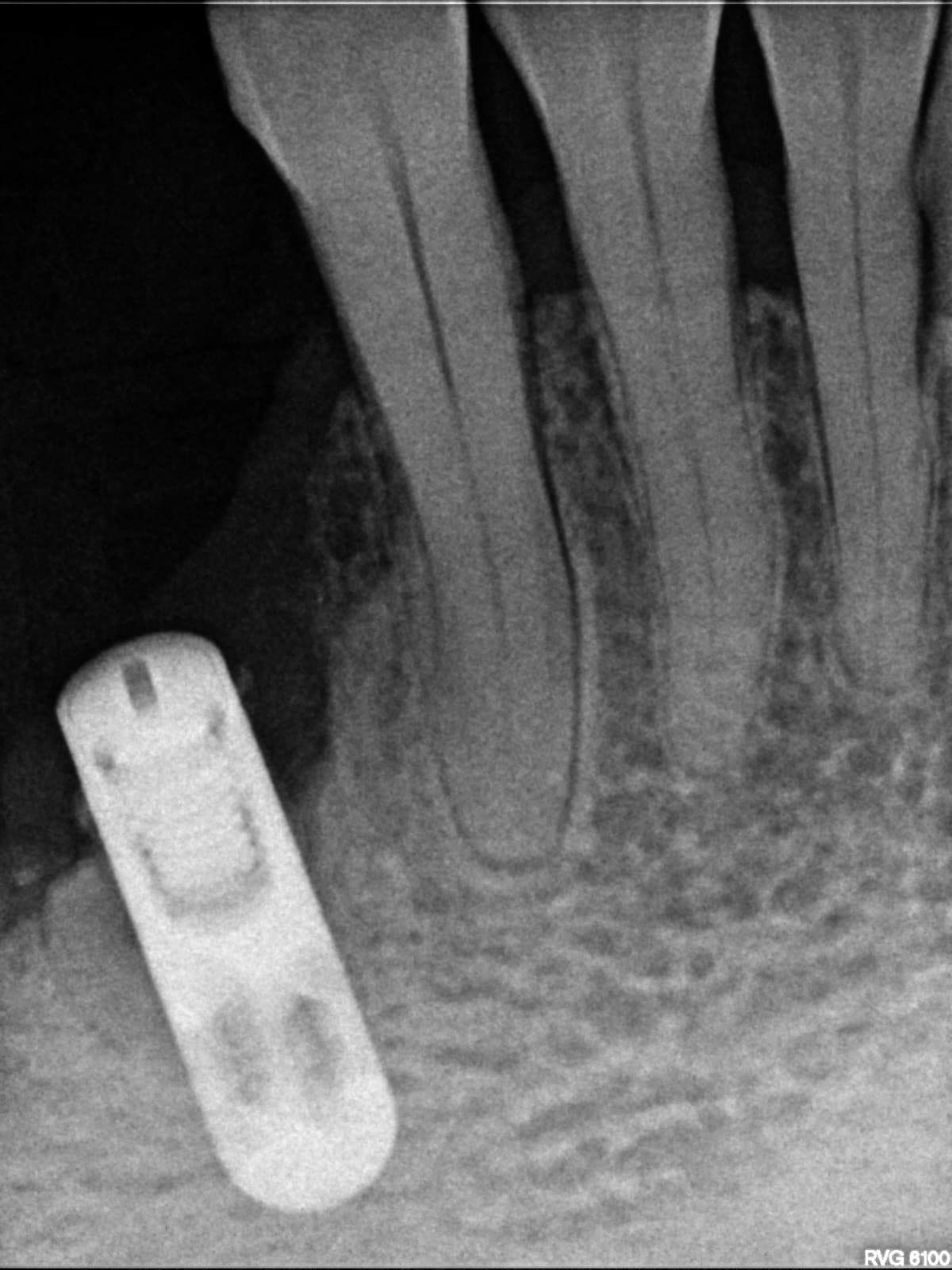
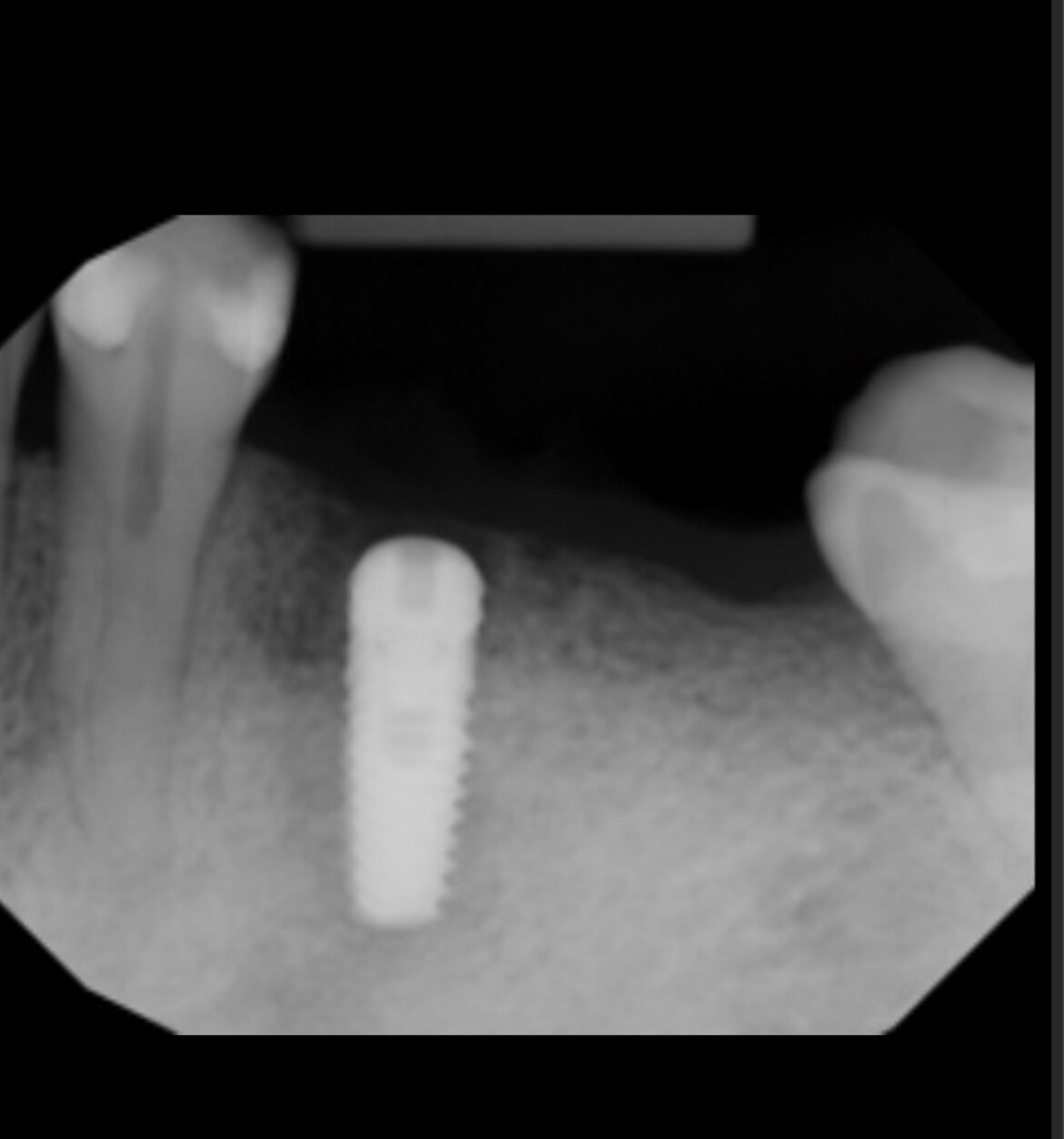
So if you’re not using an autograft, (Osteogenic,osteinductive, osteoconductive) ,all other grafts have some , rare , but failure potential. Especially if protocols like the ones Dr Moe stated Are not performed. We know the convenience of the bone in the bottle but sometimes they can fail. Xenograft especially are frowned upon here in the States( Because of their poor turnover ) and but widely used with great success in Europe. From what I was told there are some ethical issues of using allograft in Europe as well so Xenografts like Bioss are used) However, the Best European teachers like Danny Buser will scrape some autologous bone and mix it with their xenograft In order to stimulate precursor cells for osteogenesis . So poor turnover of xenograft, along with maybe as Dr Moe stated over compaction, less bleeding of socket, maybe not hydrating graft particles, maybe less than ideal primary closure w/ smoker, systemic issues w pt health , diabetics?. So many factors, When I place the graft I look at my socket and scrape it until I can see the bone and if it does not bleed I make it bleed ( your x-rays show they are endodontically treated 8;9 therefore you will get little bleeding in that socket w Endodontically treated teeth as PDL fibers are not as vital)Take a round (2-6)bur or a sharp hand instrument (curettes) and scrape ,decorticating (RAP phenomena) so blood is there , Also I prefer Allograft and recently have had great success with a quick alloplastic osteogen plug material by Implantdent . So I think you did everything correct maybe pay attention attention to bleeding , clean socket, Good closure and some good luck
After I tell you all this , on rare occasions ,I go back and I get the same result, but I think the results get more successful w experience . You did the right thing though try allograft instead of xenograft, as white xenograft particles do not remain in socket when you open , allograft turns over fast 50/50 cortico canc 250-1micron ,I use)
Hope this helps there are other clinicians who are much more learned here ,would be interested in their opinions.